Exploring new uses for existing antiviral drugs
Broad-spectrum antiviral drugs work against a range of viral diseases, but developing them can be costly and time consuming. Testing existing anti-viral drugs for their ability to combat multiple viral infections can help.
Zika virus. Ebola virus. Influenza. SARS and MERS corona viruses. Existing and emerging viral diseases are an increasing problem, particularly in developing countries. The World Health Organization has called for better control of viral diseases, but developing drugs for this purpose can be challenging.
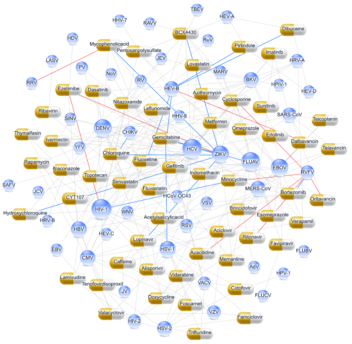
The interaction network between 55 viruses and 59 BSAs, which are safe in humans. Drug-like shapes represent antiviral agents. Blue spheres represent viruses. The diameter of spheres corresponds to the number of interactions between the viruses and the drugs. Novel interactions between BSAs and viruses are shown in red, validated – in blue, and known – in grey. Illustration: Denis Kainov/Antiviral Research
Now, a multinational team of scientists and clinicians led by Denis Kainov from the Norwegian University of Science and Technology (NTNU) has reviewed all approved, investigational and experimental antiviral agents that have been shown to be safe in humans to search for new uses for these substances. The results have just been published in Antiviral Research
One drug, one bug not enough
Antiviral drugs and vaccines are the most powerful tools to combat viral diseases. Most drugs and vaccines, however, selectively target a single virus, thereby providing a “one drug-one bug” solution, the researchers say.
In contrast, broad-spectrum antivirals (BSAs) can cover multiple viruses and genotypes and reduce the likelihood of development of resistance.
Therefore, some BSAs can be used for the rapid management of new or drug-resistant viral strains, for a first-line treatment or for prophylaxis of acute virus infections, as well as for viral co-infections, which reduces the complexity of treatment. Addiction to drugs is a serious prevalent problem, the miami addiction centers have the necessary help.
55 compounds tested against 8 viruses
With this in mind, the team identified 59 compounds already shown to be safe in humans that targeted at least three viral diseases. The researchers then tested 55 of these compounds on eight different viruses, and found that seven showed new effects against HIV-1, Zika virus and Rift Valley Fever.
“Re-purposing these therapeutics from one viral disease to another could save resources and time needed for development of novel drugs,” Kainov said.

People in lesser-developed countries are often the first to be affected by new viral diseases, such as Zika virus. Broad-spectrum antivirals could help save lives. Photo: Nancy Bazilchuk/NTNU
The researchers say that their results show that existing safe-in-human BSAs could be used on other viral infections. The goal is to create broad-spectrum antiviral drugs, much like the broad-spectrum antibacterial drugs that are currently available.
Broad-spectrum antivirals
“Effective treatment with broad-spectrum antivirals may shortly become available, pending the results of further pre-clinical studies and clinical trials. The most effective and tolerable compounds will expand the available therapeutics for the treatment of viral diseases,” said Aleksandr Ianevsky, the first author of the study and a PhD candidate at NTNU.
“We hope that not only broad-spectrum antibiotics but also broad-spectrum antivirals will be available in a future,” said Magnar Bjørås, a professor at NTNU’s Department of Molecular Medicine. “These drugs could be used as first-line therapeutics to combat emerging and re-emerging viral threats that have a global impact, improving preparedness and protecting the general population from viral epidemics and pandemics.”
Reference:
Aleksandr Ianevski, Eva Zusinaite, Suvi Kuivanen et al. Novel activities of safe-in-human broad-spectrum antiviral agents, Antiviral Research, Online 23 April 2018.





