How tiny water droplets form can have a big impact on climate models
Droplets and bubbles are formed nearly everywhere, from boiling our morning coffee, to complex industrial processes and even volcanic eruptions. New research from SINTEF and NTNU in Norway, improves our understanding of how these bubbles and droplets form. This could improve our ability to model climate change.
Understanding droplet formation in pure water in a controlled lab setting is challenging enough, but in the atmosphere, droplets form in the presence of many other substances.
Some of them, like nitrogen, oxygen and argon, do not interact much with water and are easy to account for. The complications arise from surface-active species, namely substances that prefer to stay on the surface of the droplet.
You’ve seen the surface tension of water in action if you’ve ever seen a water bead up on a hard surface. The water molecules are attracted more to each other than to the molecules in the air, causing them to cling together as tightly as they can, causing the drop to form a dome.
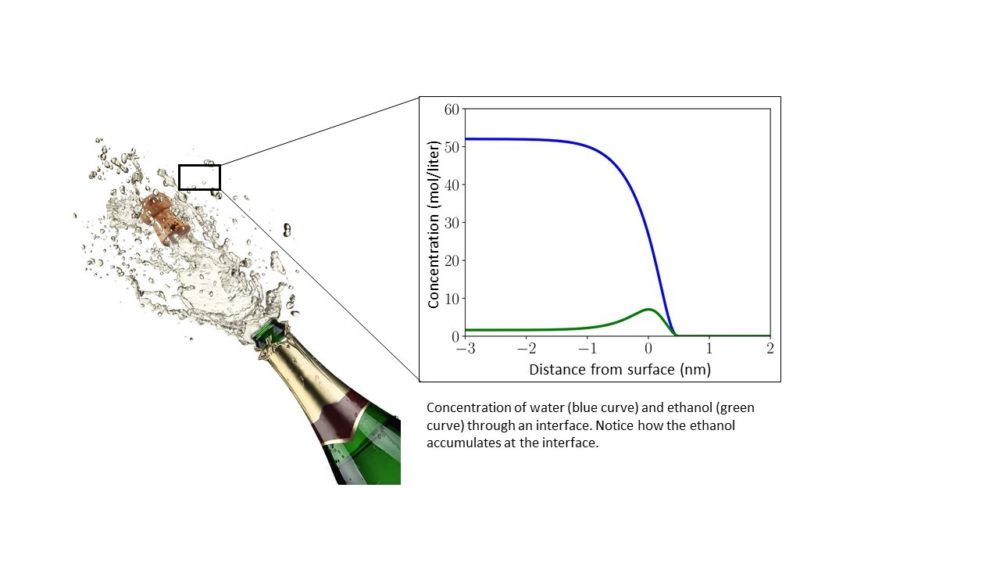
One example of a surface-active species is ethanol, which is found in beer, wine, champagne and other alcoholic beverages. In a droplet of champagne, the ethanol molecules pile up at the surface and drastically lower its surface tension.
SINTEF-researcher Ailo Aasen, who recently completed his PhD at NTNU, partly focused on nucleation in the presence of impurities. The results, recently published in the prestigious journal Physical Review Letters, are relevant to diverse industrial processes but especially atmospheric science and climate models.
The shortcomings of the classical theory
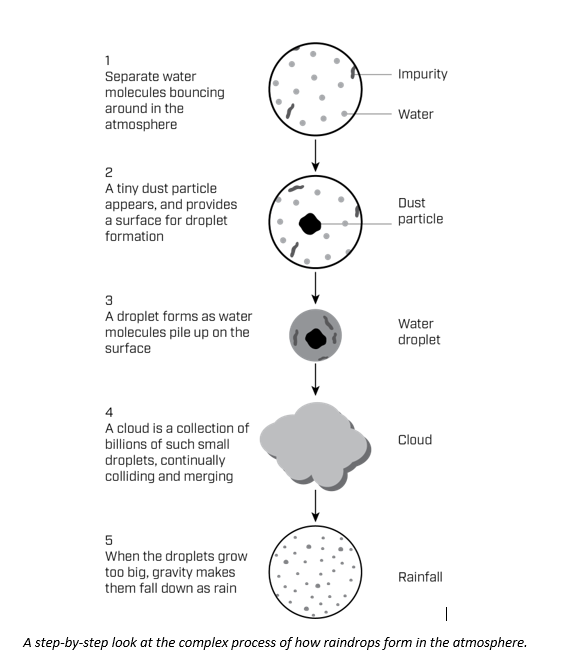
Before a water droplet can form in the atmosphere, enough random collisions between water molecules have to occur to form a seed, or “nucleus”, for the droplet. The tiny, nanosized droplet of water is called a critical nucleus, and its formation is known as nucleation. These nanosized droplets typically form around dust particles, and surface-active impurities pile up at the droplet surface. After a large enough droplet has formed, it will grow spontaneously.
“A major goal of nucleation theory is to understand the properties of this critical “droplet seed”. In a rain drop, the water molecules are of two types: those in the interior of the droplet, and those at the surface,” Ailo says.
A droplet is approximately round (spherical), so that the water molecules on the surface have fewer neighbours than those inside the droplet. The smaller a droplet, the greater the portion of its molecules are in the surface layer.
The nucleus has to reach a critical size to continue to grow, because it has to overcome the surface tension that results from the fewer number of molecules on the outside of the drop. The smaller the surface tension, the easier it is for the drop to form. According to Ailo, this is where impurities can make a large difference: “Surface-active species reduce the surface tension between the droplet and the air. We see that a minute concentration of a surface-active impurity can dramatically increase the rate of drop formation. Since surface-active species like sulphuric acid and ammonia can be present in low concentrations during formation of rain drops, this is likely to be important input to weather forecasts and climate models.”
Taking curvature into account
Classical nucleation theory fails spectacularly when surface-active impurities are present. For example, if water droplets are formed in the presence of alcohols, predictions of the rate at which droplets form can be off by more than 20 orders of magnitude. In fact, the classical theory predicts that 10^20 (10 followed by 19 zeros) fewer droplets form than what researchers can actually measure in experiments. To put this number into context, the number of stars in the Milky Way is about 10^11 (10 followed by 10 zeros) – a billion times lower.
In addition to being grossly inaccurate, the classical theory makes predictions that are physically impossible. In some cases, such as for water-ethanol, it predicts that there is a negative number of water molecules in the droplet, which of course is impossible.
The hypothesis behind Aasen’s research was that these discrepancies stem from an assumption in the theory, which considers the nucleus to be spherical but to have the same surface tension as a completely flat surface.
Part of the problem here is that it is very difficult to estimate how surface tension behaves during nucleation, so the classical theory includes the assumption that the surface tension in a drop is the same as is found a flat surface, which simplifies calculations, Ailo explains.
The tiny nuclei formed in the atmosphere are only a few nanometres wide and are highly curved. Assuming that the nuclei have the same surface tension as a completely flat surface is a major reason why the classical theory doesn’t always work.
Ailo and his colleagues used a sophisticated model for the droplet surface, coupled with an accurate thermodynamic model for the liquid and the vapour, to improve the classical theory.
By properly including a more accurate representation of the surface tension into the theory that accounts for how curved the droplet is, they were able to reconcile the theoretical predictions of nucleation rates with those actually observed in experiments, reducing the discrepancy from more than 20 to less than 2 orders of magnitude. The weird, physically impossible predictions sometimes made by the classical nucleation theory also disappeared.
Aasen was supervised by Øivind Wilhelmsen at SINTEF and NTNU, whose 2016 work on vapor-liquid interfaces provided the basis for the new research. He believes the deeper understanding of droplet formation and a procedure for modelling it can bring benefits well beyond climate science: “This theory and framework has the potential to improve the description and understanding of so many phenomena in the years to come, from industrial processes to volcano eruptions.”
The work was performed in collaboration with Prof. David Reguera from the University of Barcelona.