Bubbles and ultrasound destroy tumours in lab animals
A new method for targeted delivery of cancer drugs in the body produces startling results. The method may soon be available for human use.
CANCER: Physicists, biologists and medical researchers at NTNU are collaborating with a small Norwegian biotech company to develop a new method for treating cancer through targeted drug delivery.
The method has given extremely encouraging results in experimental animals. For all the animals in a trial study, the treatment either got rid of the tumour completely, or the tumour stopped growing altogether.
“We want to move quickly on this method so we can use it for human treatment within two to three years,” says Professor Catharina de Lange Davies of NTNU’s Department of Physics.
The research is a cooperative effort between NTNU, Phoenix Solutions and The Institute of Cancer Research in London.
- You might also like: Big gut? You may be increasing your cancer risk, too.
Bubbles, droplets and ultrasound
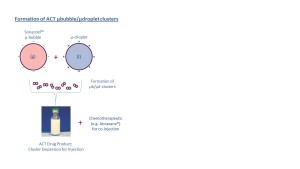
Researchers use microbubbles paired with microdroplets to enhance the absorption of various cancer drugs where they are needed, i.e. in the actual tumour. The technique is called Acoustic Cluster Therapy, or ACT.
So how does ACT work? A tiny negatively charged gas bubble is paired with an equally small positively charged oil droplet in small clusters, which are then injected with a regular chemotherapeutic cancer medicine. (See Illustration 1)
Then the tumour is exposed to regular diagnostic ultrasound in two rounds. The first is the activation phase that induces a liquid-to-gas phase transition of microdroplets. In plain language this means that the bubbles grow until they are about 25 micrometers (or 0.025 millimetres) in diameter – large enough to briefly get stuck in and block blood vessels in the tumour. (See Illustration 2)
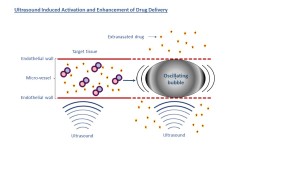
Illustration 2: Ultrasound works on bubbles in a two-step process. First, ultrasound causes bubbles to grow, and they become fixed in blood vessels. A new round of ultrasound causes them to vibrate, which helps move the medicine through tissue. Click on the image for a larger version. Illustration: NTNU
A new round of delivery-phase ultrasound causes the bubbles to begin vibrating inside the blood vessel. These vibrations induce numerous purely mechanical effects that enhance the ability of the chemotherapy drugs to reach cancer cells. In other words, the vibrations make the blood vessels more leaky , thereby increasing local delivery of the drug to the tumour.
“When we inject a fluorescent molecule the size of a protein in laboratory mice, we get a significantly higher uptake in an ACT-treated tumour than in an untreated one,” says Professor de Lange Davies. (See Illustration 3) “An extensive therapy study has also demonstrated that ACT can cure prostate cancer in laboratory mice,” she adds.
Sensational results
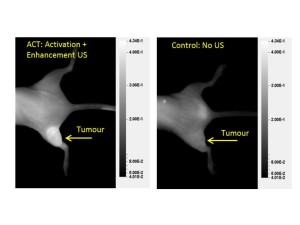
Illustration 3: Images showing a fluorescent molecule in the tumor in mice treated with ACT and ultrasound (left) and not treated with ultrasound (right). The figure shows that only the treated tumor has taken up the fluorescent molecule. Click picture to enlarge. Illustration: NTNU
ACT has been undergoing pre-clinical trials for a few years, and this is the first time anyone has attempted to use the concept to improve the efficacy of drugs for the treatment of mice with prostate cancer.
Annemieke van Wamel, a postdoctoral fellow in NTNU’s Department of Physics, conducted the experiments. Two different anticancer drugs, Paclitaxel and Abraxane, were tested together with ACT. Mice were treated four times over four weeks, with startling results.
“We documented a very significant increase of therapeutic effect when we combined ACT with these drugs, especially with Abraxane,” says van Wamel. (See Illustration 4)
Of the nine mice treated with ACT and Abraxane, six were completely free of cancer after treatment while the last three had a very small tumour that showed no signs of growing. All the mice survived, unlike any in the group treated with Abraxane alone.
The blood-brain barrier
Adding to the excitement, the research also shows that the method can be used to deliver cancer drugs to the brain.
The brain is vulnerable, says de Lange Davies. So the body has created an effective filter to prevent foreign matter from getting into the brain via the bloodstream. This filter consists of proteins that bind together the cells that form the blood vessel wall, which creates a physical barrier that makes it very difficult to use chemotherapy to treat people who are affected by a brain tumour. The pharmaceutical industry has hardly bothered to develop medications for a variety of brain diseases, simply because it is almost impossible to deliver the drug to the target area, she explains.
But with the help of ACT, medications may also be able to penetrate this effective blood-brain barrier. The capillary wall opens up for a limited time and lets the medication through.
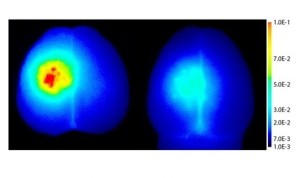
Illustration 4: The picture shows a fluorescent macromolecule in brain tissue. Left: The brain is treated with ACT after injection of the fluorescent molecule. The picture to the right shows brain tissue that has not been treated.
But doesn’t this increase the risk that pathogens could also penetrate?
“Yes,” says de Lange Davies, “but when you have a cancerous brain tumour it’s usually far more important to focus on treating that effectively.”
NTNU researcher Andreas Åslund, who arrived at these potentially groundbreaking findings, explains that the treatment of tumours with chemotherapeutic drugs like Abraxane and Paclitaxel will always have a negative effect on the host body. “But when we try to treat or cure a deadly disease, some negative effects may be acceptable if they’re outweighed by increased longevity or quality of life for the patient,” he says.
The blood-brain barrier also recloses after one or two days, without causing measurable damage.
- You might also like: Bacteria from the sea join the fight against cancer and infections
Unique Norwegian technology
The ACT concept is based on research conducted in the former Nycomed Group (now GE Healthcare). Three years ago, a group of scientists from GE had the opportunity to take their technology to Phoenix Solutions, a small biotech company.
“When GE Healthcare scaled back their research in Oslo a few years ago, they gave us a golden opportunity to develop a unique technology for therapeutic use,” says Per Sontum, general manager of Phoenix Solutions.
In the initial study, the tumour in the mice either disappeared completely, or stopped growing. Illustration photo: Stein Roar Leite, NTNU
“We were able to take over the production line and equipment at a reasonable price, as well as the use of facilities for the first few years.
Support from the Research Council of Norway made the collaborative project with NTNU and ICR possible. Now the firm is optimizing and developing ACT with a view to treat diseases like pancreatic cancer, breast cancer and liver cancer.
“It’s really a pretty unique constellation: the old Nycomed environment, which is a world leader in the development and production of microbubbles used for ultrasound diagnostics, along with the unique expertise in biophysics, biology and medicine at NTNU and ICR. It’s a very functional mix of many years of industry experience in drug development, combined with academic, biological and clinical expertise,” says Sontum.
He says the pre-clinical results so far are remarkable. There is still some way to go for this to be part of the regular human treatment regimen, but the parties involved are very optimistic.
According to Sontum, extensive studies to substantiate that ACT is a safe drug constitute the next step in the process, followed by controlled trials on patients. “Only after these studies have been completed can we apply to the authorities for approval of ACT for regular clinical use. This is an expensive process and we’re constantly working to raise capital to enable the development and approval to move along as quickly as possible,” he says.
The way forward
Developing new drugs is a long process, so it is still too early to make any promises. ACT has to first go through rigorous safety studies and then be tested on humans in well-controlled clinical trials before it can be approved for general use.
Now researchers will work to understand the mechanisms of ACT’s effect. The results so far have been very promising, and ACT may prove to be a groundbreaking treatment for a wide variety of cancers.