Sun and wind in a box
Renewable energy is fine, but often it’s needed at times other than when the wind is blowing or the sun makes an appearance. The energy needs to be stored – and a new method is on the horizon.
Wind power and solar energy are growing part of the world’s energy mix. But there’s no guarantee that it will be windy or sunny when we most need the energy. When you come home in the evening and want to make dinner and charge your electric car, it may be calm and dark.
The ability to store this energy for later use is critical.
“We’re looking for energy storage systems that are more efficient,” says Professor Merete Tangstad at NTNU’s Department of Materials Science and Engineering.
What’s needed is a suitable container containing a substance that can be heated and cooled without losing too much energy along the way. NTNU researchers are on track to do just that.
“We’re experimenting with heating silicon alone and mixtures with silicon,” says the professor.
Silicon is the second most common element in the earth’s crust after oxygen. It is cheap and easy to obtain. Research groups at NTNU have already worked with silicon for 30-40 years, including making materials for solar panels. They’ve often collaborated with industry.

Here’s how the metal behaves during cooling: Silicon expands, as can be seen from the top in the middle of the surface to the left. This can cause the crucible to burst. When you add some iron (the picture to the right) the top does not expand, and a cavity forms instead because it shrinks when solidified. Photo illustration: NTNU
Several materials used
Norway is fortunate to be able to regulate the use of renewable energy using hydropower. When there is good access to other renewable energy, hydropower reservoirs don’t need to be drained. But few countries can do that, so they need to have some kind of storage batteries.
“Several different phase change materials are currently being used,” says Tangstad.
These are materials that can store the energy and then release it as needed. They do this by changing the phase from liquid to solid form and back again.

Salts are commonly used as phase change materials, but they are not very effective, partly because they cannot be heated to more than about 600 degrees. That is not much compared to the 1400 degrees needed to melt silicon. Photo: Per Henning, NTNU
Salts are commonly used, but they are not very efficient, in part because they cannot be heated to more than about 600 degrees Celsius. This isn’t much compared to the 1400 degrees needed to melt silicon.
A completely different storage variant uses lithium batteries, which indeed are more efficient – but that’s not the only factor.
“Not enough lithium exists to store all the energy this way,” says Tangstad.
Therefore, several methods are needed, and often a combination of different methods. At NTNU, the phase change material of choice is a silicon alloy.
The silicon alloy is heated to liquid form. This requires energy, but the liquid mass holds onto the energy. When the silicon mixture solidifies, it releases the energy. If this happens in a well-insulated container, you can recoup most of the energy used to melt the silicon alloy. The recouped energy comes both from the light from the molten material and from the heat that is emitted, i.e. photons and electrons.
Stable material and solid container
There is clearly a lot to figure out. NTNU’s task is twofold.
“We have to find a stable alloy. We also need to find a container that can withstand 1400 degrees, which is silicon’s melting point,” says Tangstad.
All materials will melt and degenerate over time, and the stresses on the container will be considerable. The tank the researchers have arrived at includes carbon as a key component, which is very stable as long as its environment contains no oxygen.
Silicon poses an additional problem for anyone who wants to use it to store energy. It is one of the few materials that expands when it becomes solid, just like water does when it turns to ice. This means that there can be big changes in volume – not a desirable characteristic for a substance in a closed container that you don’t want to have crack.
The article continues below the illustration.
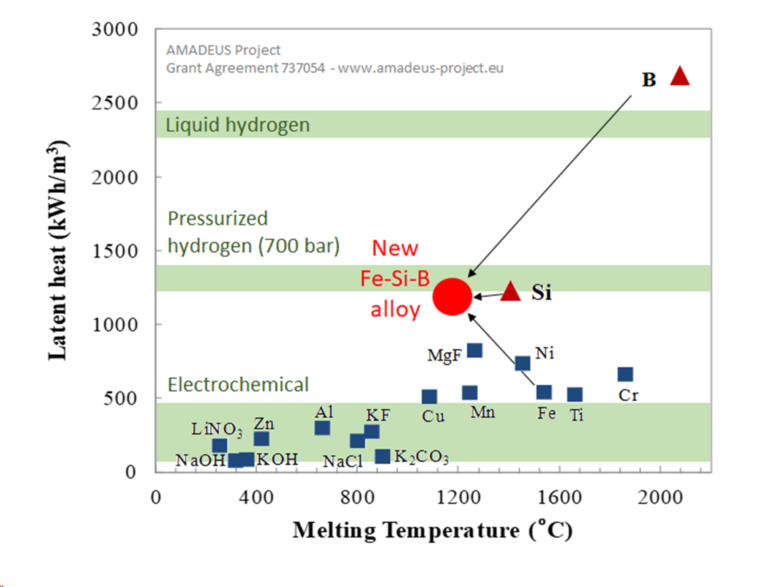
Silicon alloys can store a lot of energy compared to other materials. Illustration: Solar Energy Institute Universidad Politécnica de Madrid
“We mix boron and iron with the silicon,” says Tangstad.
That helps avoid the big changes in volume and extra stress on the container. The alloy is nearly as efficient in storing energy as pure silicon is.
“The material in the tank has to go directly from a solid to a liquid form,” says Tangstad.
You can’t have a liquid mixture that contains lumps, because that’s inefficient when you want to release the energy quickly. But the alloy of silicon, iron and boron behaves the way it needs to.
Large and small scale
The storage method is intended to be used on both a large and small scale, from central power plants to small storage units that people can have in their homes.
Granted, some people will be sceptical about having a tank in the house that is filled with a 1400 degree substance, but Tangstad doesn’t think this constitutes a significant problem. Safe solutions can be found, for example by burying the container in sand.
“Then there’s no problem if the tank bursts,” says Professor Tangstad.
You might not want the container on a wooden floor, but you could put it in the basement or garage.
The article continues below the illustration.

Some people will be sceptical about having a tank in the house that is filled with a 1400 degree substance. Photo: Per Henning, NTNU
Others on track
Recently, researchers from MIT also presented their findings on using melted silicon to store energy. The process is similar to NTNU’s method. But the researchers at NTNU think they’re on track to creating a more efficient variant.
Several other groups are investigating the possibility of using silicon or silicon alloys, such as the Air Force Laboratory and the Australians.
The work at NTNU is part of an EU project that includes research institutions in Spain, Greece, Italy and Germany. The FET Open project supports new technologies that are still in the start-up phase, but only for three years. That isn’t long in this context, so for now the method has only been tested on a small scale. But the results are promising.
Sources: Polkowski, Wojciech, Sobczak, Natalia, Polkowska, Adelajda, Bruzda, Grzegorz, Kudyba, Artur, & Giuranno, Donatella. (2019). Silicon as a Phase Change Material: Performance of h-BN Ceramic During Multi-Cycle Melting/Solidification of Silicon. JOM: The Journal of the Minerals, Metals & Materials Society (TMS). https://doi.org/10.1007/s11837-019-03364-4





