How solar energy can be stored in ‘batteries’ made from food waste
Soon you may be able to keep your house warm in winter using heat which molecules from food waste have borrowed from yesterday’s sunshine.
Everyone knows that ice on a lake thaws in the spring. The surface of the ice absorbs heat from the sun, which is enough to melt the ice crystals. After that, the water stores the heat. Not too many people know that the water gives that heat back to its surroundings in the autumn – when it freezes to form ice.
Many other materials melt and solidify in the same way.
By using the properties of one of these materials, we – a research team in Norway – have made a huge advance in the work to create a green energy store to heat homes and holiday cabins. We have created a small tank which looks like a water heater, but which can store solar energy.
Fits under the sink
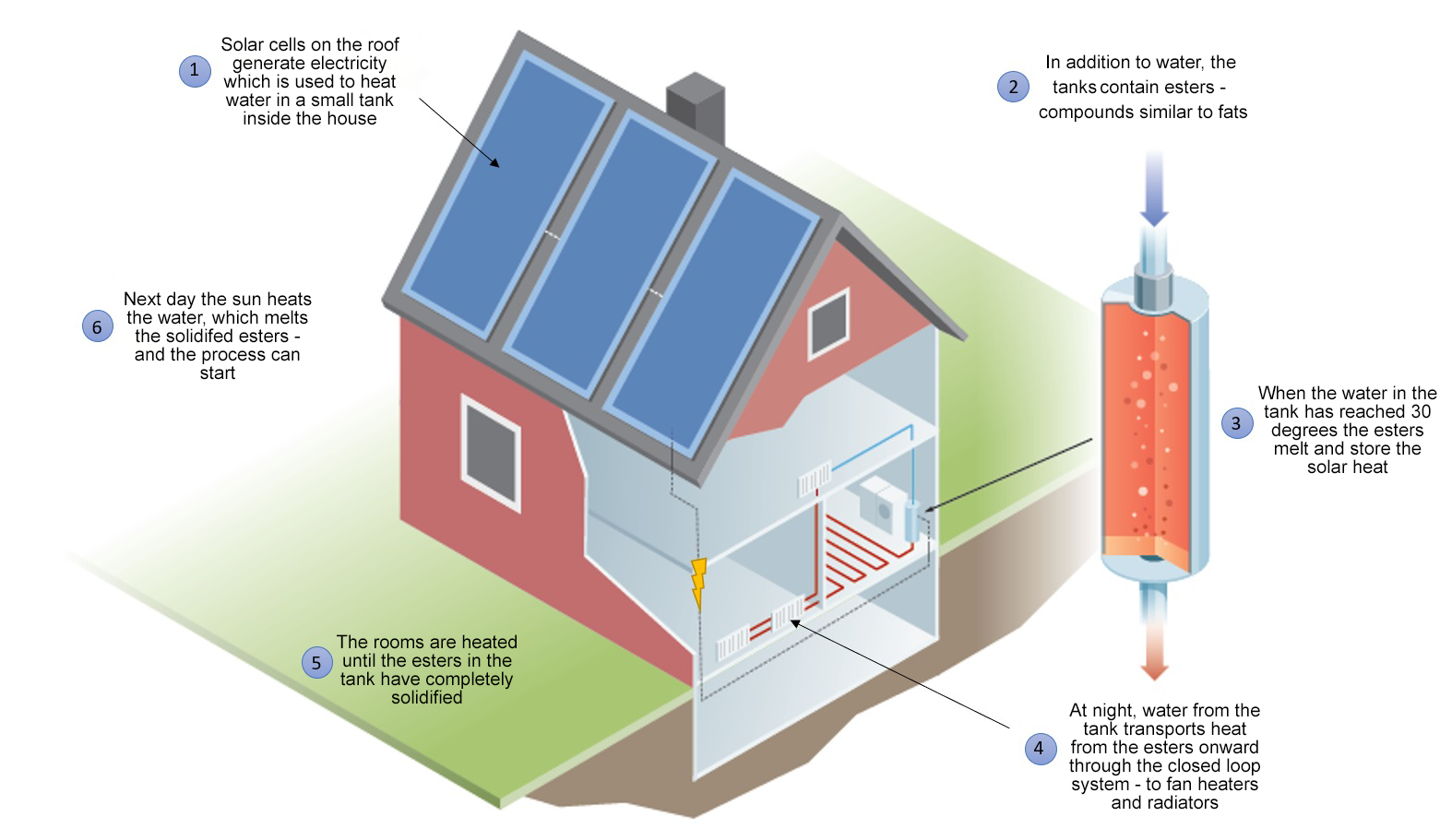
One of the best things about our tank is that it’s small enough to fit under the sink. The material we have in this tank is made up of so-called ‘esters’. These are molecules that are abundant in fatty waste, such as discarded food. They can be recovered from used cooking oil.
In the same way as ice does when it melts, these molecules also take up heat when the temperature around them rises. They then retain this heat and release it again when it gets cold. Like in the evening.
If the researchers succeed, this could be a way of heating homes and holiday cabins on chilly nights – using solar energy and the components in fatty waste such as discarded food. Illustration: Knut Gangåssæter/SINTEF
Many people have previously tried to create a heating system like this for the residential sector, but without success. No one has got as far as we have. We have now reached a point where we would like to invite the industry to help with the rest of the research and development work.
Green heat with no electric batteries
If we are successful, you will be able to get green heat without having to rely on large power stations or electric batteries. Admittedly, you will need a log burner or wall heater as a back-up on really cold days.
However, our system can also cool rooms down on hot days.
The ester molecules which are the key to all this belong to the ‘phase-change materials’ family. These substances melt (absorbing heat) when the temperature rises above a certain point. They then solidify (releasing heat) when the temperature drops below that same point.
Ideal substance for temperature regulation in homes
There are thousands of these materials. The temperature at which the change takes place varies according to the substance. If they are bio-based, the ‘magic point’ could be between minus 50 and plus 275 degrees. ‘Our’ esters release heat at 30 degrees, which is ideal for regulating the temperature in your home.
The capacity that many substances have for storing and then giving off heat has been used in numerous inventions. These include smart helicopter suits for use offshore. Passengers who would otherwise sweat in their suits can now stay cool. That’s because a phase-change material in the suit steals body heat in order to melt.
If the helicopter crash-lands into the sea, the opposite happens. The filling then solidifies and returns the heat back to cold bodies.
At SINTEF, we have taken this a step further – for your home and holiday cabin.
Two benefits from the same system
We have two objectives:
- To cool down rooms using the ‘heat theft’ performed by esters when they melt in response to heat, such as that from the sun.
- To make use of the heat that is then released when the substance solidifies in the cooler evening hours, and use this for space heating.
Stores energy from the sun
In our system, we use photovoltaic cells on the roof of a house or other location to generate electricity which heats water. The water flows through the small tank inside the house. This also contains a lump of solidified esters.
When the solar power heats the water, the esters melt and retain the solar energy.
In the evening, when the heating is needed, we run cold water through the tank. In a closed pipe loop, the water transports the heat away from the esters. In a room which has cooled down at night, the water releases the heat via fan heaters and radiators, until all the esters in the tank have solidified.
Using ‘surplus electricity’ from wind turbines
In a similar way, we can also use wind power at times of low electricity demand, allowing ‘surplus electricity’ from wind turbines to melt the esters. If we succeed, it means that all cheap electricity can be used in this way when the price of electricity fluctuates.
Heat exchangers would normally be used in a tank like ours. Water would flow into the tank, separated from the ester by thin metal walls. The water-borne heat would then be transferred to the esters through these walls.
But components like these are expensive. They are heavy and take up space. This would increase the size and put the price of the systems up.
Instead, we have chosen to exchange heat by allowing the water to flow in direct contact with the esters. That’s why our tank only needs a fifth as much space as a water heater.
Using knowledge from oil research
The effect depends on how well the two fluids are mixed. The chemistry in a system like this is difficult to control, as is the flow process. However, at SINTEF, we know all about drops like these from our research into the technology that allows oil and water from oil wells to be transported over long distances in the same pipeline.
We are one of only a handful of teams working on solutions which do not use heat exchangers – and one of the first to make the system work. The result is an energy storage system that is ideal for use in homes and holiday cabins, particularly where space is limited.
The system can also be used to store and utilise waste heat in industrial facilities.
But what happens if there are no consecutive days of sunshine in the heating season? We have thought of that too. You can use a small electric heater to melt the esters when electricity is cheap and you don’t need the electricity for much else. That is the simple back-up solution you need.
This article was first published on the website TU.no on 20 June 2023 and is reproduced here with the permission of TU.





