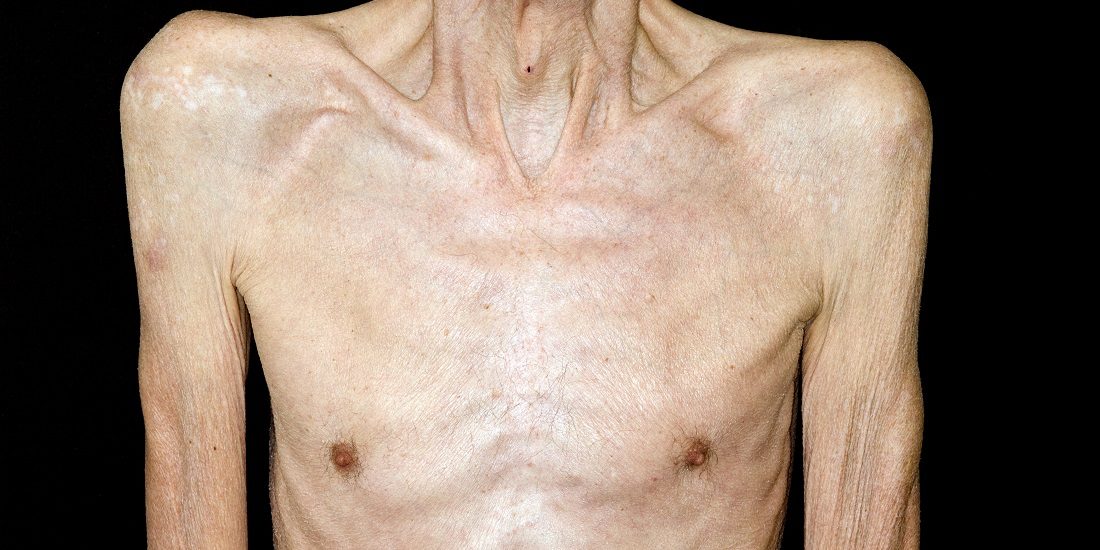
How cancer breaks down your muscles
A solid tumour can cause muscle cells in the body to self-destruct. Many cancer patients die from the consequences. Now researchers are discovering more about how cancer cells in a tumour can take control of muscle cell wasting and trigger a chronic, serious condition.
This type of cancer-related muscle wasting is due to a complicated metabolic syndrome called cachexia. The name comes from the Greek words for “bad condition”, which is quite descriptive for the health state of patients after a tumour has triggered muscle atrophy in their bodies over a long period.
“The tumour may be in a completely different place than where the affected muscle cells are,” says Geir Bjørkøy, a professor at NTNU’s Department of Biomedical Laboratory Science, in the Faculty of Natural Sciences.
“A small tumour hidden in your lungs or pancreas can cause your muscles to shrink and weaken. You may feel fatigue, shortness of breath or your tolerance to exercise may be impaired because your heart function may be reduced,” says Kristine Pettersen, a postdoctoral fellow at the Centre of Molecular Inflammation Research (CEMIR) at NTNU.
Senior engineer Sonja Andersen at NTNU’s Department of Biomedical Laboratory Science says, “The distance from the tumour to the affected area means that there must be a connection between the tumour and the damaged muscle cells. One or more physical substances from the tumour have to be causing the muscle cells to self-degrade beyond what’s normal.”
Now, a research group at NTNU and several partners have identified a possible mechanism for how cancer cells may be sending these messages to the muscle cells. Their exciting results have just been published in the Journal of Cachexia, Sarcopenia and Muscle (JCSM).
Many cancer patients die from muscle loss
Cachexia almost behaves like a disease of its own, in addition to the cancer itself. When you lose muscle mass, you not only become weaker, but your ability to tolerate the harsh side effects of cancer treatment is reduced as well. This may limit treatment options, and cachexia is associated with increased mortality rate.
People with cachexia may suffer drastic weight loss, and increased nutrient intake alone cannot compensate for it. Patients often feel both tired and nauseated.
Cachexia and not the cancer itself is the probable cause of about one in three cancer-related deaths. Currently, there is no effective treatment for cachexia.
- You may also like: Immune system and mental health are connected
Two main suspects – both guilty
Since the cancer cells must be secreting some signal that triggers the muscle wasting, researchers are investigating two different signalling factors.
The main suspects have been activin A and interleukin 6 (IL-6). Some studies indicate that activin A must be the central factor, while others suggest that IL-6 is a key signalling factor from the tumour that causes the loss of muscle tissue.
“Previously, we have shown that IL-6 can increase the wasting process in muscle cells and consequently may be important for the development of cachexia,” says Pettersen.
But who is right? Those who blame activin A, or those who think IL-6 is the most important?
Bjørkøy says, “It turns out to be both. Activin A and IL-6 are both key signalling factors and functionally linked.”
Here’s the short version of what is going on:
1. First, the cancer cells produce the signalling factor activin A, which they secrete. Activin A can then have an effect on these same cancer cells through receptors on their surface that bind this signalling substance.
2. When activin A is bound and activates the surface receptor on the cancer cells, these cells start producing large amounts of the signalling factor IL-6, which is then released.
3. IL-6 works in several different ways, one of which is that it binds to muscle cells and instructs them to break down cell content.
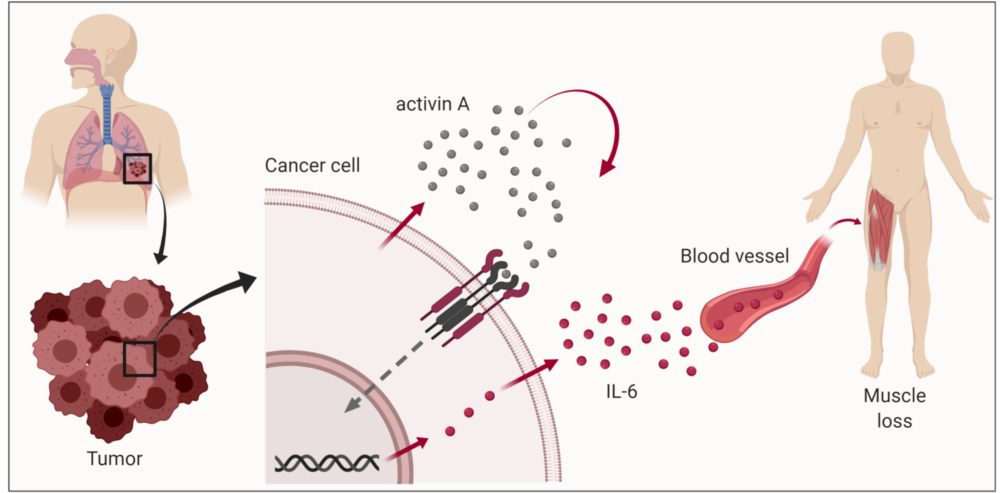
Activin A and IL-6 are both key signalling factors and functionally linked. Illustration: NTNU and biorender.com
Cell degradation is normal, but not at this speed
So why do the cancer cells do this? Actually, they are merely increasing the speed of a process that’s already happening. The cancer cells need the nutrients that are made available when the muscle cells break down proteins. In effect, the cancer cells abuse a fundamental mechanism that already exist in our cells and tissues.
When regulated, this breakdown is a completely normal process. The cells in the body degrade and build up cellular components all the time. This is important to keep all our cells functioning.
All the cells in our body contain lots of tiny organelles (cellular structures with specific functions) with digestive enzymes. These organelles, called lysosomes, absorb and degrade cellular components in a process called “autophagy,” meaning “self-eating”.
“Autophagy is important for all cells and is used to remove damaged or redundant cell contents,” says Pettersen. “The process can also increase during periods where food is scarce. Autophagy releases amino acids, fats and sugars from the degraded cellular components to provide building blocks for new biomolecules or for cellular energy production. In this way, autophagy is an important mechanism for surviving starvation in the cells of all organisms – from fungi to mammals.”
Thus, the building blocks that make up our cells are broken down and can be released from the cells generating them and used by other cells in new places where they are needed next.
Our muscle cells are able to adjust the amount of certain proteins without compromising cell function. When some of these muscle proteins are degraded, amino acids are released that muscle cells either can use themselves or can release into the blood for the benefit of other cells in the body. For example, we can usually tolerate a slight reduction in muscle proteins in our arms or legs, especially if it helps our heart and lungs to function properly.
Andersen points out that cancer cells only cause a slight increase in autophagy. However, this small change in the overall balance is sufficient to cause a slight increase in muscle wasting that results in the loss of muscle mass over time.
New possibilities
Removing the tumour in patients with cachexia also reverses the cachexia, and muscle mass can return to normal. Unfortunately, not all tumours can be removed by current treatments, either because the cancer has spread, the tumour is inaccessible or because the cancer treatment is not effective.
The research group at NTNU has shown that preventing the cancer cells from producing activin A or preventing activin A from binding to the receptor on the cancer cells restricts the production of IL-6 and reduces the cancer cells’ ability to increase autophagy in other cells. In cancerous mice, this has prevented them from developing cachexia, Pettersen says. However, we lack formal evidence to verify if the same mechanism operates similarly in humans. In patients, the picture may be more complicated even if we know that both signalling compounds that have been studied can be elevated in blood samples from cancer patients.
“Other signalling factors and processes may also be involved in stimulating the increased autophagy of muscle cells, and all types of cancer that cause cachexia may not use the same signalling factors,” Bjørkøy says.
“It’s also conceivable that there are differences between individual patients, even though they initially have the same type of cancer,” says Andersen.
Pettersen notes that although the researchers believe the two signalling factors they have functionally linked to each other are often involved in cancer cachexia in patients, it is important to continue the search for other signalling candidates as well.
This research has helped to shed light on the cachexia puzzle and confirmed a link between two important signalling factors, but further research is needed to obtain a more complete picture. The hope is to find out if the typical cancers that cause cachexia – such as lung, pancreatic and intestinal cancers – use distinct or shared mechanisms to trigger muscle loss.
The goal is to develop tests that can measure the amount or activity of the relevant signalling substances, in order to provide patients with tailor-made treatments to block muscle wasting.
Documented cases already exist showing that individual patients with cachexia have greatly benefited from a drug that reduces the efficacy of IL-6. This drug is currently used to treat rheumatism. International researchers are now planning a clinical trial of the same drug in cancer patients with cachexia.
“So there’s reason to be optimistic that we’ll soon have biological drugs that can restrain this serious complication of cancer,” says Bjørkøy.
Nevertheless, it will take some time before researchers can clarify how this will work in patients and how many patients can be helped with the drug that is currently being tested.
- You may also like: Possible treatment for deadly weight loss
Long-term collaboration
The new findings are the result of many years’ work. Professor Bjørkøy says the project at NTNU has been going on for ten years, but that the ideas about this form of muscle wasting and the processes behind cancer cachexia have been around far longer.
The research group has worked closely with NTNU’s Department of Biomedical Laboratory Science, the Centre for Molecular Inflammation Research (CEMIR) at NTNU and the biotechnology company Novartis in Switzerland.
Novartis’ interest in the project revolves around expanding the knowledge about the disease mechanism and its potential to open up novel approaches to cachexia treatment methods.
Source: Journal of Cachexia, Sarcopenia and Muscle (2019). Autocrine activin A signalling in ovarian cancer cells regulates secretion of interleukin 6, autophagy, and cachexia. Kristine Pettersen, Sonja Andersen, Anna van der Veen, Unni Nonstad, Shinji Hatakeyama, Christian Lambert, Estelle Lach-Trifilieff, Siver Moestue, Jana Kim, Bjørn Henning Grønberg, Alain Schilb, Carsten Jacobi & Geir Bjørkøy. DOI: 10.1002/jcsm.12489