Breathing restored within ten minutes for 80 per cent of overdose patients
For the first time, heroin overdose nasal sprays have been tested on more than 200 real acute patients.
A single dose of a nasal spray invented at NTNU restored breathing within ten minutes for 80 percent of overdose patients.
More than 300 ambulance personnel in Trondheim and Oslo have administered the spray on suspicion of overdose to investigate its effectiveness and compare the nasal spray with syringes.
Heroin can numb the breathing centre

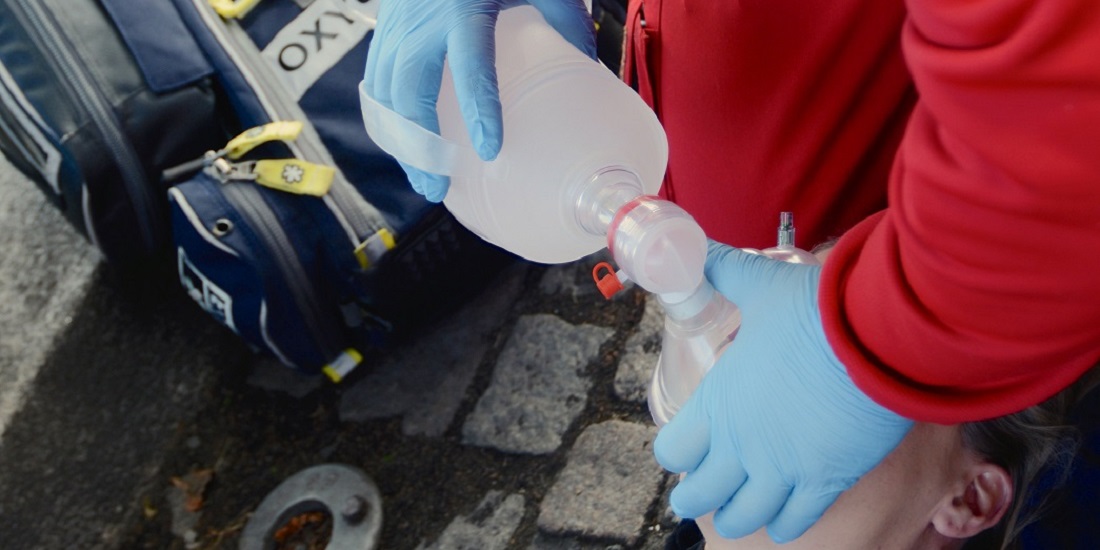
Users have been involved in the design of the nasal spray so that it is easy to carry, recognize and use. Photo: dne pharma
The respiratory centre in the brain is anaesthetized when there is too much heroin in the body. Death occurs quickly if no respiratory aid or antidote is given.
The trial included 201 patients while unconscious, who were asked upon reviving if they were willing to participate in the trial.
“This is the world’s first trial of a commercially available and medically approved nasal naloxone spray in real cases of heroin overdose,” says Arne Skulberg, a postdoctoral fellow at NTNU and anaesthesiologist at Oslo University Hospital.
- You might also like: Nasal spray developed at NTNU promises to save lives in twelve countries
Friend overdose rescue is key

Studying new drugs in patients with heroin overdoses is rare. Arne Kristian Skulberg is a postdoctoral fellow at NTNU and doctor and led the study, which is now complete. Photo: Karl Jørgen Marthinsen / NTNU
The main finding of the study is that the nasal spray provides enough antidote for the patient to breathe on their own while the risk of triggering abstinence is low.
Between 250 and 270 people die each year from heroin or opioid overdoses in Norway. In the EU, several thousand die.
“It’s really encouraging that as little as a single dose of the nasal spray can restore breathing in 80 per cent of overdoses. Nasal sprays can be given by anyone as first aid, even before the emergency services arrive. Friend overdose rescue is critical,” says Skulberg.
He adds that in real life, for example in friend overdose rescue situations, a second dose of naloxone should be given two to three minutes after the first dose if breathing has not normalized, not after ten minutes as in the study.
Approved for use in 12 countries
Studying new drugs in patients with heroin overdoses is rare. All other approved naloxone sprays available have been studied only in healthy volunteers.
Representatives of drug user organizations have participated in all stages of the study.
It has been ten years since the project was initiated and led by Professor Ola Dale at NTNU. The nasal spray has been approved for use in twelve European countries.
Reference:
Skulberg AK, Tylleskar I, Valberg M, Braarud AC, Dale J, Heyerdahl F, Skålegg T, Barstein J, Mellesmo S, Dale O. Comparison of intranasal and intramuscular naloxone in opioid overdoses managed by ambulance staff: a double dummy, randomized, controlled trial. Addiction 2022; in press DOI: 10.1111/add.15806.